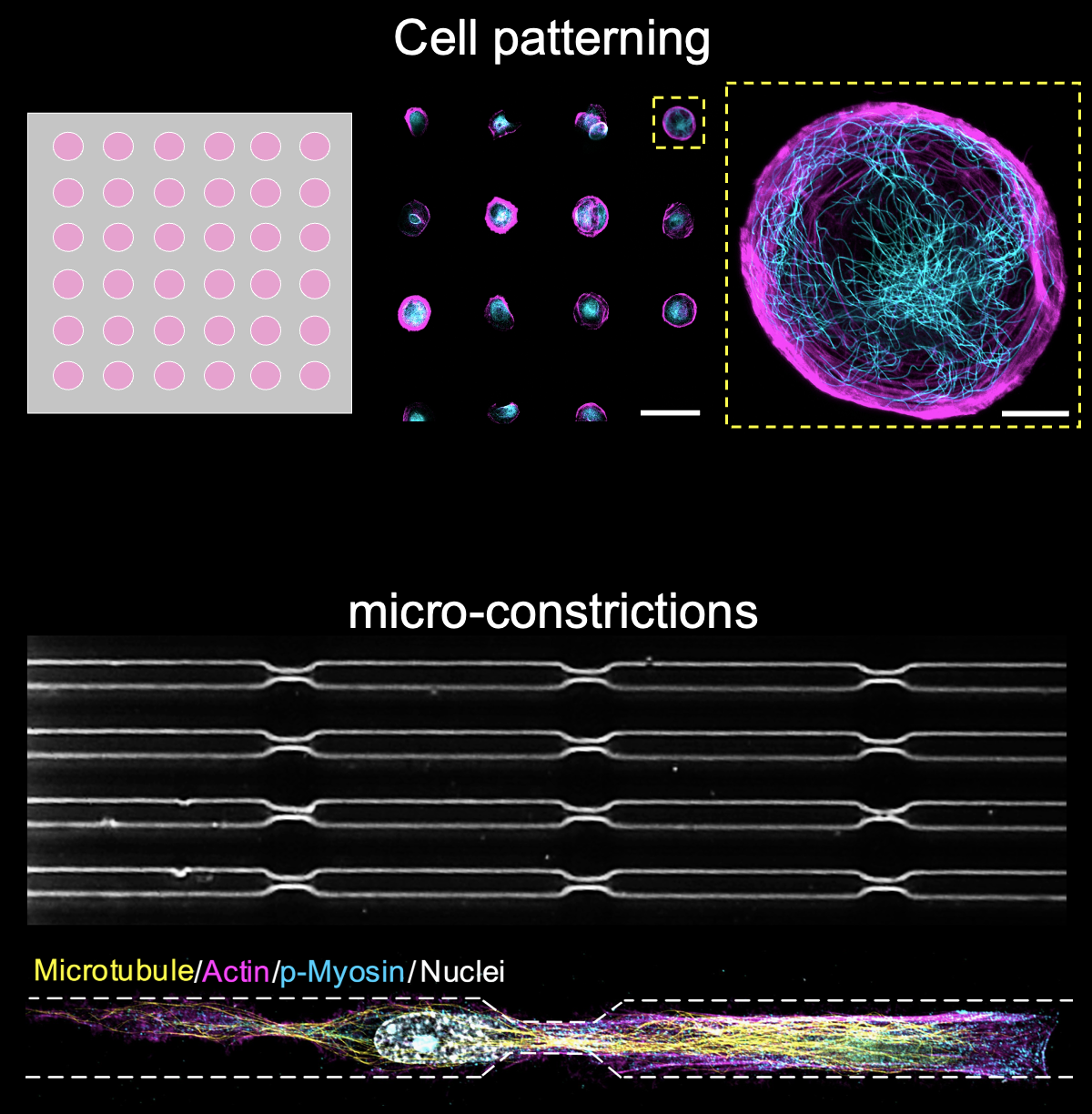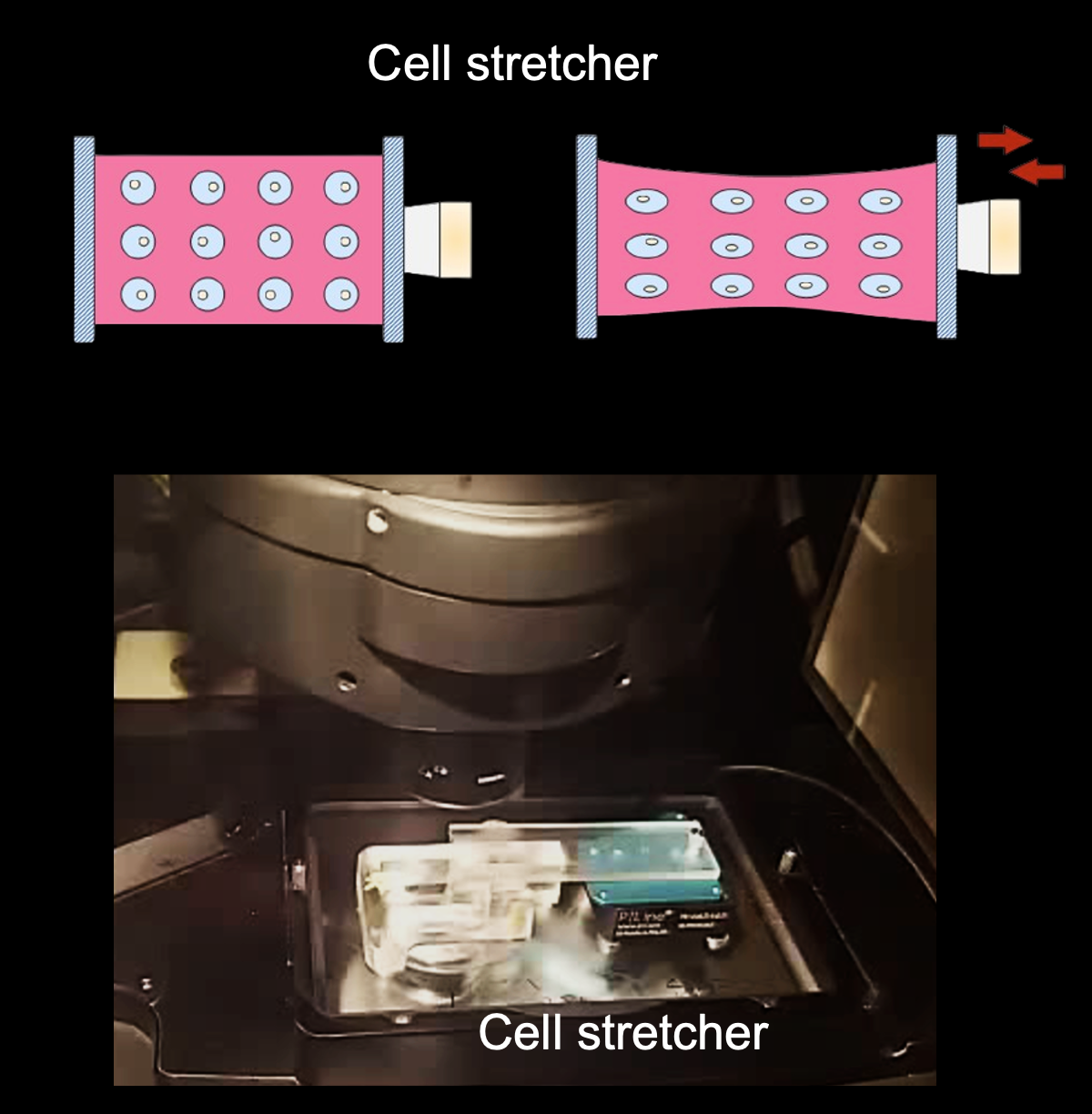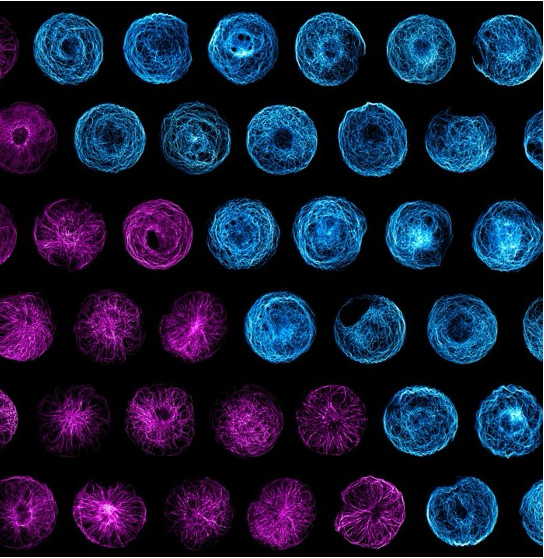
Microtubule organisation and mechanosensation
Microtubules (MTs) are stiff and dynamic polymers spanning the cytoplasm in mammalian cells. They are essential for maintaining vital living activities including mass transport, cell division, polarisation and ciliation. Our previous work (Nature Materials 2023) reported that the microtubule network can not only bear mechanical forces but rather respond to them, functioning as a microscale mechanosensor in living cells. MTs increase their stability to resist external forces by relocating plus-tip proteins along their entire length, and such a mechano-response is essential when cells migrate through constricted spaces. We are particularly interested in how MT mechanosensation coordinates with other cytoskeletons (e.g., actin and intermediate filaments) in cell migration or organoid morphogenesis models.